A solution usually boils at a higher point than the pure solvent. By increasing the air pressure above the water Apex Jacob V.
The greater the pressure the more energy required for liquids to boil and the higher the boiling point.

. AnswerExplanationAdding any solute to water will increase the boiling temperature as it reduces the vapour pressure meaning a slightly higher temperature is Search 1. How do you increase the boiling point of water. Sugar salt or other non-volatile solutes.
How do you increase the boiling point of a liquid. Additionally changing the pressure on a liquid changes its boiling point. The boiling point of a liquid varies depending upon the surrounding environmental pressure.
A liquid in a partial vacuum has a lower boiling point than when that liquid is at atmospheric pressure. The addition of solutes or other substances usually changes the boiling point. The pressure of gas above a liquid affects the boiling point.
This is because the presence of the solute decreases the vapour pressure of the solution. The addition of solutes or other substances usually changes the boiling point. Additionally changing the pressure on a liquid changes its boiling point.
How do you increase the boiling point of water. In an open system this is called atmospheric pressure. The greater the pressure the more energy required for liquids to boil and the higher the boiling point.
Sugar salt or other non-volatile solutes in water will usually make the boiling point higher. Increasing the external pressure also raises the boiling point. Adding any solute to water will increase the boiling temperature as it reduces the vapour pressure meaning a slightly higher temperature is required in order for the vapour pressure to become equal to atmospheric pressure and boil the water.
In an open system this is called atmospheric pressure. An effective way of raising the boiling point of a liquid is to add another ingredient. See answer 1 Best Answer.
Boiling points can be changed in several ways. Atmospheric Pressure and Boiling. Atmospheric Pressure and Boiling The pressure of gas above a liquid affects the boiling point.
Boiling points can be changed in several ways. While water at sea level has a boiling point of 100 degrees Celsius its boiling point can be raised by adding a solute such as salt. You can increase the pressure above the boiling water.
Adding a salt generally increases the bp. A liquid at high pressure has a higher boiling point than when that liquid is at.

Thermal Performances Of A Flat Plate Pulsating Heat Pipe Tested With Water Aqueous Mixtures And Surfactants Sciencedirect
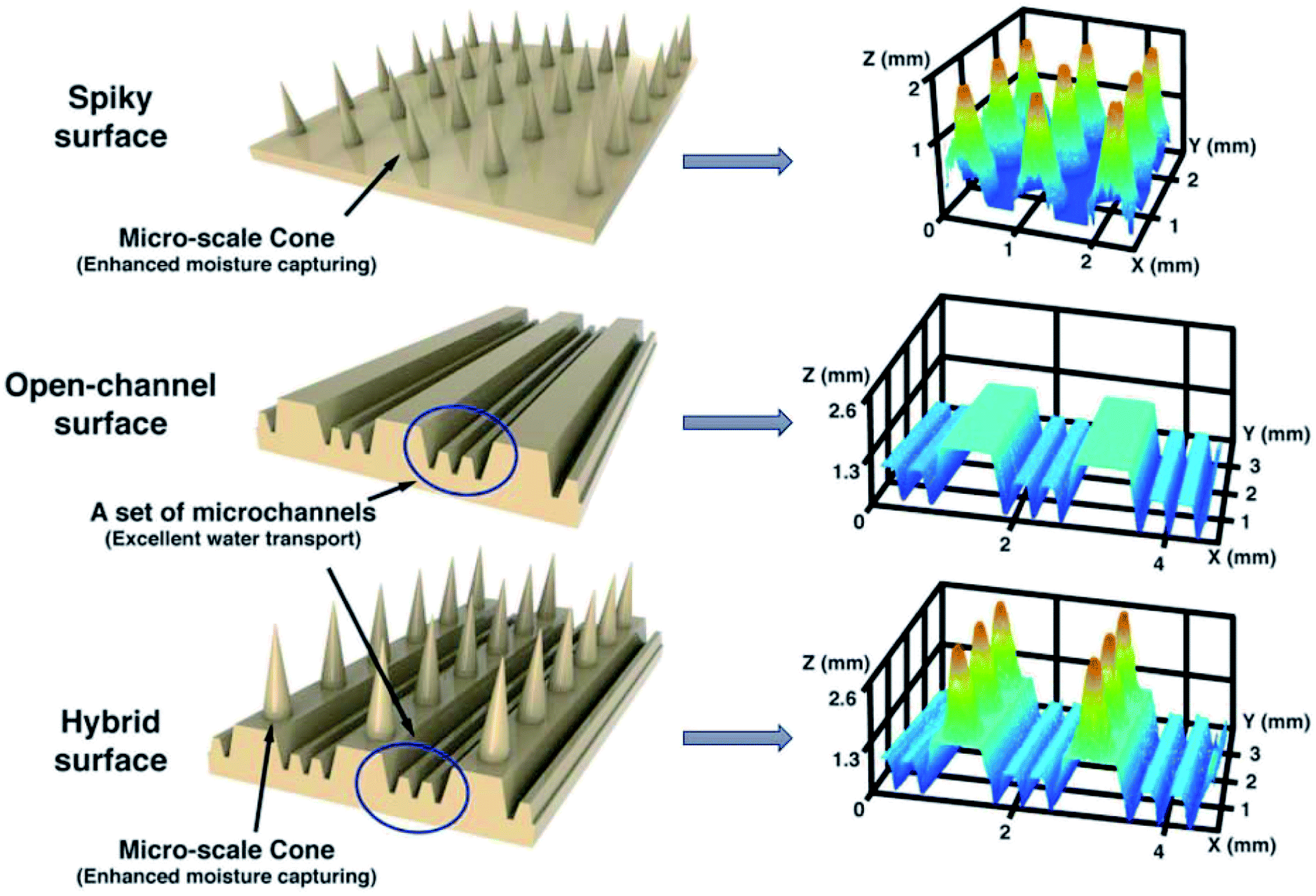
The Intrigue Of Directional Water Collection Interface Mechanisms And Strategies Journal Of Materials Chemistry A Rsc Publishing Doi 10 1039 D1ta06182h

Quarrtz Floor Standing Bottled Water Cooler The Water Cooler Company

0 Comments